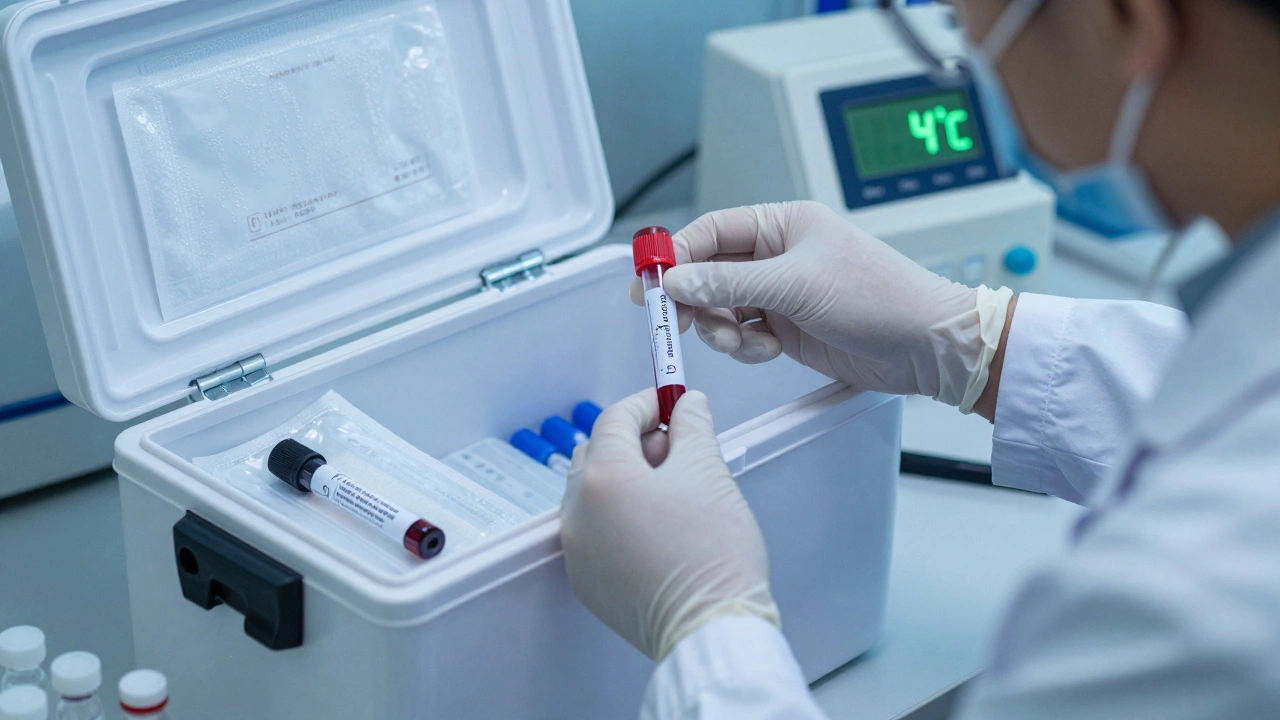
When a blood sample, tissue biopsy, or DNA swab leaves the collection site, its journey doesn’t end there. What happens next-how long it sits, how cold it gets, how many times it’s frozen and thawed-can make or break an entire investigation, clinical test, or research study. Temperature and timing aren’t just logistical details; they’re the foundation of sample integrity. Get this wrong, and even the most carefully collected sample becomes useless.
Why Temperature Matters More Than You Think
Biological samples don’t just sit still. They’re alive in a chemical sense, even after collection. Enzymes keep breaking down proteins. RNA unravels. Cells burst. Bacteria multiply. If you don’t control the environment, degradation starts the moment the sample leaves the body. The CDC and NIST agree on three main temperature zones for custody:- Room temperature (15-25°C): Only for fixed or preserved samples, like paraffin-embedded tissues or alcohol-stored specimens. Even then, DNA degrades over time. RNA? Gone within hours.
- Refrigerated (2-8°C): The sweet spot for short-term holding. Think 24-72 hours max. This keeps enzymes sluggish without freezing cells. Ideal for blood, urine, or swabs headed to the lab within a day or two.
- Frozen (-20°C and below): This is where long-term storage begins. But not all freezing is equal.
Here’s the catch: freezing at -20°C isn’t enough for many samples. If you’re storing stem cells, reproductive tissue, or clinical trial samples, you’re looking at -80°C or colder. At -20°C, ice crystals still form slowly and can rupture cell membranes. At -80°C, that damage slows to a crawl. Below -135°C? Biological activity essentially stops.
Timing: How Long Can You Wait?
Temperature alone doesn’t tell the whole story. Time is equally critical.- Whole blood: Must be refrigerated within 30 minutes of collection. If left at room temp longer than 4 hours, white blood cells start dying, and cell-free DNA levels rise unnaturally-messing up tests for cancer or prenatal screening.
- RNA samples: Degrade fast. Even at 4°C, RNA half-life is under 24 hours. If you’re doing gene expression analysis, freeze within 2 hours. No exceptions.
- Tissue biopsies: If you’re preserving for histology, formalin fixation buys you days. But if you need molecular analysis, flash-freeze in liquid nitrogen within minutes. Waiting 30 minutes can reduce RNA quality by over 70%.
- Sperm and embryos: These aren’t just stored-they’re cryopreserved. Delaying freezing by even an hour can drop viability by 20% or more.
There’s no universal clock. Each sample type has its own deadline. The rule? When in doubt, freeze sooner. Once degradation starts, you can’t reverse it.
Freeze-Thaw Cycles: The Silent Killer
One of the biggest mistakes labs and evidence rooms make? Repeatedly thawing and refreezing samples. Every time a sample warms up-even to room temperature for 10 minutes-ice crystals melt and reform. This tears apart cell membranes, denatures proteins, and fragments DNA. A sample that’s been thawed three times may have 80% less usable DNA than one frozen once and kept cold. The fix? Aliquotting. Instead of pulling out the whole tube every time you need a drop of serum or tissue lysate, divide the sample into small, single-use portions before freezing. Label them clearly. Use only what you need. Never return thawed material to the freezer.And when you do thaw? Do it fast. A water bath at 37°C takes 2-3 minutes. A fridge thaw? That’s 2-3 hours-and during that time, degradation is happening.

Storage Equipment: Not All Freezers Are Created Equal
You can’t just stick samples in any old freezer and call it good.- Frost-free freezers: These cycle heat every few hours to melt ice. That means your sample hits 0°C twice a day. Bad. Avoid them entirely for biological storage.
- Standard -20°C freezers: Fine for some reagents, but not for long-term sample preservation. Temperature can drift ±3°C. Not precise enough for research-grade work.
- Ultra-low freezers (-80°C): The minimum for most biobanks. Must have alarms, backup power, and temperature logging. Check logs weekly.
- Cryogenic storage (-150°C to -190°C): Used for irreplaceable samples like donor stem cells, rare tissue archives, or clinical trial biologics. Requires liquid nitrogen or mechanical cryo-systems. No power outages allowed.
Thermo Fisher, Azenta, and BioLife Solutions all agree: if you’re storing samples for more than 6 months, assume you need -80°C or colder. If it’s worth keeping, it’s worth freezing right.
Sample-Specific Rules
Different materials need different rules. Here’s a quick guide:| Sample Type | Optimal Temperature | Max Storage Time | Key Notes |
|---|---|---|---|
| Whole Blood (EDTA) | 2-8°C | 48 hours | Freeze at -80°C for DNA/RNA testing |
| Plasma/Serum | -20°C | 1 year | Aliquot to avoid freeze-thaw |
| RNA (e.g., from swabs) | -80°C | 5+ years | Freeze within 2 hours of collection |
| DNA (saliva, blood) | -20°C | 10+ years | Stable, but -80°C preferred for long-term |
| Tissue Biopsy (unfixed) | -135°C | Indefinite | Flash-freeze in liquid nitrogen |
| Sperm / Embryos | -190°C | Indefinite | Require controlled-rate freezing |
| Bacteria (culture) | -80°C | 5+ years | Use glycerol stock, not plain freeze |
Shipping and Chain of Custody
Temperature control doesn’t end at the lab door. Shipping is where most samples fail.- Use validated cold packs-not ice. Ice melts and can leak, contaminating samples.
- For refrigerated samples, ensure the package stays between 2-8°C during transit. Use temperature loggers.
- For frozen samples, use dry ice. But don’t pack it directly against the sample. Wrap it in cardboard or foam. Direct contact can freeze and crack tubes.
- Always document the temperature at each handoff. Signature logs, timestamped data, and barcodes aren’t optional-they’re legal requirements in forensics and clinical trials.
One study in forensic labs found that 37% of DNA samples submitted for analysis were compromised due to improper shipping. That’s not just bad science-it’s a risk to justice.
What Happens When You Get It Wrong?
Bad storage doesn’t just mean "inconclusive results." It means:- False negatives in cancer screening because RNA degraded.
- Wrong paternity results because DNA was fragmented.
- Failed clinical trials because the control group samples were unusable.
- Legal cases dismissed because evidence was mishandled.
There’s no second chance. Once a sample degrades, you can’t recover it. No test can fix what the cold never protected.
Best Practices Summary
- Know your sample. RNA? Freeze fast. Tissue? Flash-freeze. Blood? Refrigerate then freeze.
- Never reuse thawed samples. Always aliquot.
- Use -80°C for anything stored longer than 6 months.
- Use cryogenic storage for irreplaceable samples (stem cells, embryos, rare tissues).
- Log temperature at every step-from collection to analysis.
- Train everyone who handles samples. One mistake by one person can ruin months of work.
Biological samples are fragile. They don’t care if you’re busy, tired, or in a rush. They only care if you kept them cold, kept them dry, and kept them frozen once.
Can I store biological samples in a regular home freezer?
No. Home freezers are not reliable. They cycle temperature to prevent frost, often hitting 0°C multiple times a day. This damages cells and degrades DNA and RNA. Even if the display says -18°C, the actual temperature inside fluctuates. Always use a lab-grade or medical freezer with temperature logging.
How long can I keep a blood sample at room temperature before it’s ruined?
For most molecular tests, blood should be refrigerated within 30 minutes. After 4 hours, white blood cells begin to break down, releasing DNA that shouldn’t be there-skewing results. For DNA testing, you might get usable material up to 24 hours, but it’s unreliable. For RNA or gene expression tests, anything over 2 hours is unusable.
Why can’t I just freeze samples at -20°C and forget about them?
-20°C works for short-term storage of some samples, like plasma or DNA, but it’s not enough for long-term preservation. Ice crystals form slowly at this temperature and can rupture cell membranes. For tissue, stem cells, or reproductive samples, -20°C will cause irreversible damage over months. Use -80°C or cryogenic storage for anything you plan to keep longer than a year.
Is it okay to thaw a sample and then re-freeze it?
Never. Each freeze-thaw cycle fragments DNA, denatures proteins, and kills cells. Even one cycle can reduce sample quality by 30-50%. Always divide your sample into small portions before freezing. Use only what you need. Never return thawed material to the freezer.
What’s the best way to transport biological samples?
Use validated shipping containers with phase-change materials or dry ice, depending on the required temperature. Never use regular ice-it melts and leaks. Include temperature loggers that record data during transit. Always document who handled the sample and when. For forensic samples, chain-of-custody forms are legally required. For clinical samples, follow FDA or CLIA guidelines.
Do I need to use special containers for freezing samples?
Yes. Use cryo-vials or tubes made for low temperatures. Regular plastic tubes can crack at -80°C. Always use screw-cap tubes with silicone gaskets-they seal better than snap-top lids. Label everything clearly with waterproof ink. Store samples in racks designed for ultra-low temps to avoid accidental spills or temperature spikes when opening the freezer.