Most people confuse quality control with quality assurance, but in a lab setting, mixing them up can cost you your accreditation. While assurance is the big-picture plan-the policies, the training, and the design-quality control is the actual boots-on-the-ground work. It is the specific set of measurements and tests you run to prove that your results are accurate and that your process hasn't drifted into error. If QA is the map, QC is the GPS telling you exactly where you are in real-time.
The Foundation of Lab Verification
You can't just start testing and hope for the best. Before a single sample is collected, you need data quality objectives. These are the benchmarks that define what "success" looks like for a specific test. Without these, you're just guessing.
To maintain a gold-standard lab, every process must follow these rules:
- Standardized Procedures: No "ad hoc" methods. Every step must be documented in a Standard Operating Procedure (SOP) so that any qualified technician gets the same result.
- Sample Integrity: From the moment a sample is collected to the second it's analyzed, it must be secured. A single contaminated vial or a temperature spike in a fridge can invalidate a week's worth of work.
- Traceability: If an auditor asks where a specific result came from three years ago, you need a paper or digital trail that connects the final report back to the raw sample and the specific machine used.
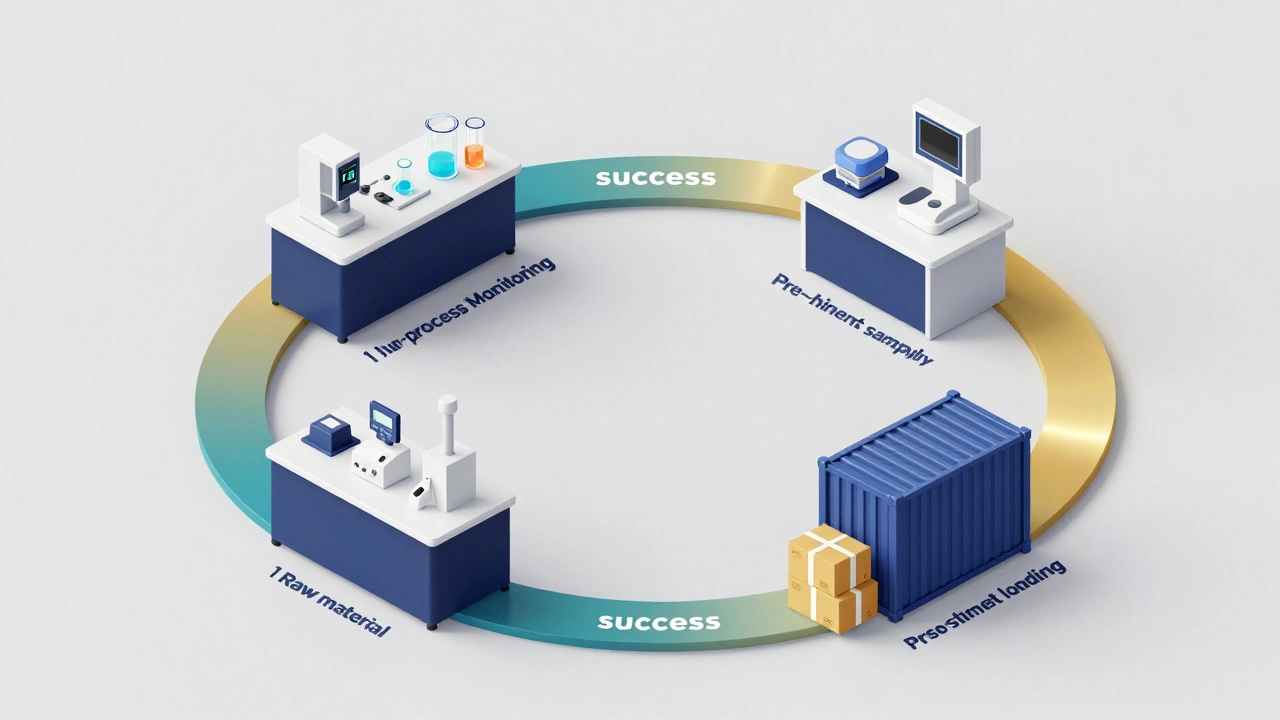
When to Inspect: The Four Stages of QC
Quality isn't a final checkbox; it's a continuous loop. In a production or lab environment, inspections happen at specific intervals to catch errors before they become expensive disasters.
First, there's the pre-production phase. This is where you check your raw materials and reagents. If your chemicals are expired or your equipment isn't calibrated, the rest of the process is a waste of time. Then comes in-process inspection. This happens while the work is moving. By monitoring a process in real-time, you can catch a drift in accuracy and fix it before you finish a whole batch of samples.
As you reach the end, the Pre-Shipment Inspection (PSI) kicks in. Usually, this happens when about 80% of an order is packed. You pull random samples and test them against the buyer's specifications. Finally, there's the container loading inspection, which ensures that the packaging is secure and the quantities are correct.
For high-value or high-risk items, some labs use piece-by-piece inspection. This is 100% verification where every single item is checked for function and safety. It's slow, but it's the only way to guarantee zero defects.
| Stage | Timing | Primary Goal | Risk of Skipping |
|---|---|---|---|
| Pre-production | Before start | Verify raw materials | Fundamental process failure |
| In-process | During production | Detect early drifts | Massive batch rework |
| Pre-shipment | 80% completion | Final spec compliance | Customer rejection |
| Loading | Final packaging | Quantity & Condition | Shipping damage/Shortages |
Technical Methods for Finding Defects
Depending on what you're testing, your methods will vary. Visual inspection is the most basic-looking for inconsistencies or deviations. But humans get tired and miss things, which is why automated testing is so critical. Machines don't blink, and they can check strength, size, and alignment with a level of precision that humans can't match.
For those seeking accreditation, Quality Audits are the heartbeat of the system. These are systematic reviews conducted by internal or external experts. They don't just look at the product; they look at the records, the facility, and the compliance with safety regulations.
When things go wrong, you need more than a quick fix. This is where Root Cause Analysis (RCA) comes in. Instead of just treating the symptom, RCA uses tools like the "5 Whys" or Fishbone diagrams to find the actual source of the failure. If a machine is producing errors, the symptom is the error, but the root cause might be a worn-out bearing or an untrained operator.
For complex systems, labs use the Design of Experiments (DOE). This is a structured way to test multiple variables at once. Instead of changing one thing at a time, DOE helps you find the optimal combination of settings to maximize quality and minimize defects.
The Science of Sampling
You can't test every single molecule in a batch-it's too expensive and often destructive. That's why sampling is used. But you can't just pick samples at random; you need a statistical basis.
The ISO 2859-1 standard, often referred to as AQL (Acceptable Quality Limit), provides a framework for this. AQL helps a lab manager determine the maximum number of defective units that are acceptable in a batch before the entire lot must be rejected. This balances cost-effectiveness with a statistically valid level of confidence in the results.
Specialized Testing for Software and Digital Systems
In modern labs, the software controlling the instruments is just as important as the hardware. Software QC is split into two main worlds: static and dynamic testing.
Static testing is a "non-execution" phase. This involves code reviews and walkthroughs where experts scrutinize the source code for logic errors before the program ever runs. It's essentially proofreading for programmers.
Dynamic testing, however, requires the software to actually run. This includes:
- Black Box Testing: The tester focuses only on inputs and outputs. They don't care how the code works, only that if they put in "X," they get "Y." This is common for user acceptance tests.
- White Box Testing: The tester has full visibility of the internal code and tests the internal paths and logic.
- Exploratory Testing: A more flexible approach where the tester "probes" the system to find edge-case bugs that a scripted test might miss.

Connecting QC to Final Delivery
The end goal of all these procedures is a feedback loop. When a quality inspector finds a defect during a pre-shipment check, that information shouldn't just stop at a rejected report. It needs to go back to the production team immediately.
This loop ensures that the lab isn't just catching mistakes, but preventing them from happening again. When you have a tight link between the inspector and the operator, you move from a reactive state (fixing errors) to a proactive state (optimizing the process). This is the difference between a lab that barely passes an audit and one that leads its field in reliability.
What is the main difference between Quality Assurance and Quality Control?
Quality Assurance (QA) is the broad, preventative framework that defines the standards, processes, and training. Quality Control (QC) is the reactive, detective part of the process that uses specific tests and inspections to verify that those standards were actually met in the final product.
Why is ISO 2859-1 important for lab testing?
ISO 2859-1, or AQL, allows labs to use statistical sampling rather than testing 100% of a batch. It provides a scientifically valid way to determine the sample size and the number of defects that can be tolerated before a batch is rejected, ensuring quality without wasting resources.
What is the "5 Whys" technique in Root Cause Analysis?
The "5 Whys" is a problem-solving method where you ask "Why?" repeatedly (usually five times) until you move past the symptoms of a problem and find the actual root cause. For example, if a sample is contaminated, you don't just clean the vial; you ask why it was contaminated, then why the seal failed, then why the seal was old, until you find the procurement error that allowed old seals to be used.
When should a lab use piece-by-piece inspection?
Piece-by-piece inspection (100% inspection) is recommended for extremely high-value items, critical safety components, or when the cost of a single defect reaching the customer is catastrophically high. It is not practical for high-volume, low-cost items.
What is the difference between Black Box and White Box testing?
Black Box testing examines the software's functionality from the outside without knowing the internal code, focusing on inputs and outputs. White Box testing involves examining the internal logic and structure of the code itself to ensure all internal paths are functioning correctly.
Next Steps for Lab Managers
If you're preparing for accreditation, start by mapping your current workflow against the four inspection stages mentioned above. Identify where you have "blind spots"-those gaps where a sample could be compromised without anyone noticing. Focus on tightening your recordkeeping and traceability first; an auditor will forgive a minor technical error if you can prove exactly how it happened, but they won't forgive a missing logbook.





